Correlation analysis of Poor Prognosis and Immunotherapy of lncRNAs Related with m6A Modification in Cervical Cancer
-
摘要:目的 本研究旨在通过挖掘宫颈癌TCGA数据库,分析N6-甲基腺嘌呤(N6-methyladenosine, m6A)修饰相关长链非编码RNA(lncRNA)与宫颈癌预后不良及免疫治疗的相关性,从而有效地评估宫颈癌患者的预后和宫颈癌免疫治疗的可行性。方法 基于TCGA数据库宫颈癌样本,利用生物信息学的方法鉴定与宫颈癌预后相关的m6A修饰相关lncRNA,并以此构建宫颈癌的预后风险模型。结果 从304例患者样本中筛选出343个m6A修饰相关lncRNA,通过单因素Cox回归分析得到26个m6A修饰相关lncRNA与宫颈癌患者预后相关,并利用Lasso回归分析得到7个m6A修饰相关lncRNA(DLEU1、AC099850.4、DDN-AS1、EP300-AS1、AC131159.1、AL441992.2、AL021707.6)用以构建预后风险模型。Kaplan-Meier曲线显示低风险组的OS高于高风险组(P<0.001);ROC曲线下面积(AUC)表明本风险模型准确性高、可信度强;多因素Cox分析显示风险评分是评估宫颈癌患者预后的独立因素。TIDE评分预测高风险组接受免疫治疗后获益更大。免疫检查点PD1与DDN-AS1等m6A修饰相关lncRNA表达相关,且在高风险组中表达更高(P<0.05)。结论 基于上述7个m6A修饰相关lncRNA构建的预后风险模型能够有效预测宫颈癌患者的预后,并能评估以PD1为靶点的免疫治疗疗效。Abstract:Objective To study the correlation between N6-methyladenosine (m6A)-modification-associated long non-coding RNAs (lncRNAs) and poor prognosis and immunotherapy in cervical cancer based on data mining of The Cancer Genome Atlas (TCGA) cervical cancer dataset, so as to assess effectively the prognosis of cervical cancer patients and the feasibility of immunotherapy.Methods We identified m6A-modification-associated lncRNAs correlated to the prognosis of cervical cancer by conducting bioinformatics analysis of cervical cancer samples from the TCGA datasets and constructed a prognostic risk model of cervical cancer accordingly.Results A total of 343 m6A-modification-associated lncRNAs were identified from the samples of 304 cervical cancer patients. Univariate Cox regression analysis showed that 26 out of the 343 m6A-modification-associated lncRNAs were significantly associated with the prognosis of cervical cancer patients. We identified 7 m6A-modification-associated lncRNAs, including DLEU1, AC099850.4, DDN-AS1, EP300-AS1, AC131159.1, AL441992.2, and AL021707.6 through Lasso regression analysis and then developed a prognostic risk model based on them. According to the Kaplan-Meier survival analysis, cervical cancer patients in the low-risk group exhibited significantly improved overall survival (OS) in comparison with those in the high-risk group (P<0.001). The area under the curve (AUC) of receiver operating characteristic (ROC) curve analysis demonstrated the high sensitivity and credibility of the risk model. Multivariate Cox analysis showed that the risk score was an independent prognostic factor of cervical cancer patients. Tumor immune dysfunction and exclusion (TIDE) analysis predicted that the high-risk group would benefit more from immunotherapy. In addition, we found that immune checkpoint PD1 was associated with the expression of m6A-modification-related lncRNAs such as DDN-AS1, and the expression was higher in the high-risk group (P<0.05).Conclusion The prognostic risk model constructed on the basis of the aforementioned 7 m6A-modification-associated lncRNAs can be used to effectively predict the prognosis of cervical cancer patients and assess the efficacy of immunotherapy targeting PD1.
-
Keywords:
- Cervical cancer /
- m6A methylation /
- lncRNA /
- Prognosis /
- Immunotherapy
-
据《2018全球癌症统计报告》,宫颈癌是女性发病率排名第四位的恶性肿瘤,其死亡病例占7.7%[1]。目前对于临床Ⅰ~Ⅱ期未发生淋巴结转移的宫颈癌患者,其主要的治疗方式是手术联合放疗或化疗;而对于临床Ⅲ~Ⅳ期患者以及已经发生淋巴结转移的Ⅰ~Ⅱ期患者来说,治愈率较低,预后较差[2]。即使经过治疗,部分宫颈癌患者仍然会出现远处转移和局部复发[3]。研究表明免疫治疗能够显著延长晚期肿瘤患者的中位生存期,同时减少复发和转移的发生[4],因此治疗前评估患者预后及免疫治疗疗效具有重要意义。
长链非编码RNA(long non-coding RNAs, lncRNA)是一种长度大于200个核苷酸的非编码RNA,能够通过参与肿瘤的生长转移[5]、恶性转化[6]和肿瘤微环境的动态变化[7]等过程影响肿瘤的发生发展。N6-甲基腺嘌呤(N6-methyladenosine, m6A)修饰是指在甲基转移酶的作用下催化RNA的腺嘌呤(A)的第六位氮原子(N)发生甲基化[8],涉及甲基转移酶催化m6A甲基化,结合蛋白识别底物RNA,去甲基化酶去除m6A甲基化等过程的动态调控[9-10]。m6A修饰通过作用于lncRNA与特定DNA位点之间的RNA-DNA三螺旋结构进而调控lncRNA的功能[11]。lncRNA LINRIS通过与结合蛋白IGF2BP2泛素化位点结合促进结直肠癌的增殖,且其高表达与结直肠癌患者预后不良正相关[12]。此外,甲基转移酶METTL3通过m6A甲基化HK2 mRNA促进宫颈癌发生发展[13]。
本研究基于TCGA数据库鉴定出7个具有宫颈癌预后意义的m6A修饰相关lncRNA(DLEU1、EP300-AS1、AC131159.1、AC099850.4、DDN-AS1、AL441992.2、AL021707.6)并构建预后风险模型,通过Kaplan-Meier(KM)曲线和ROC曲线证实该模型的预测能力,同时评估以PD1作为宫颈癌免疫治疗靶点的可行性,为以PD1作为宫颈癌有效的免疫治疗靶点提供了预测指标和理论依据。
1. 资料与方法
1.1 数据的提取与整理
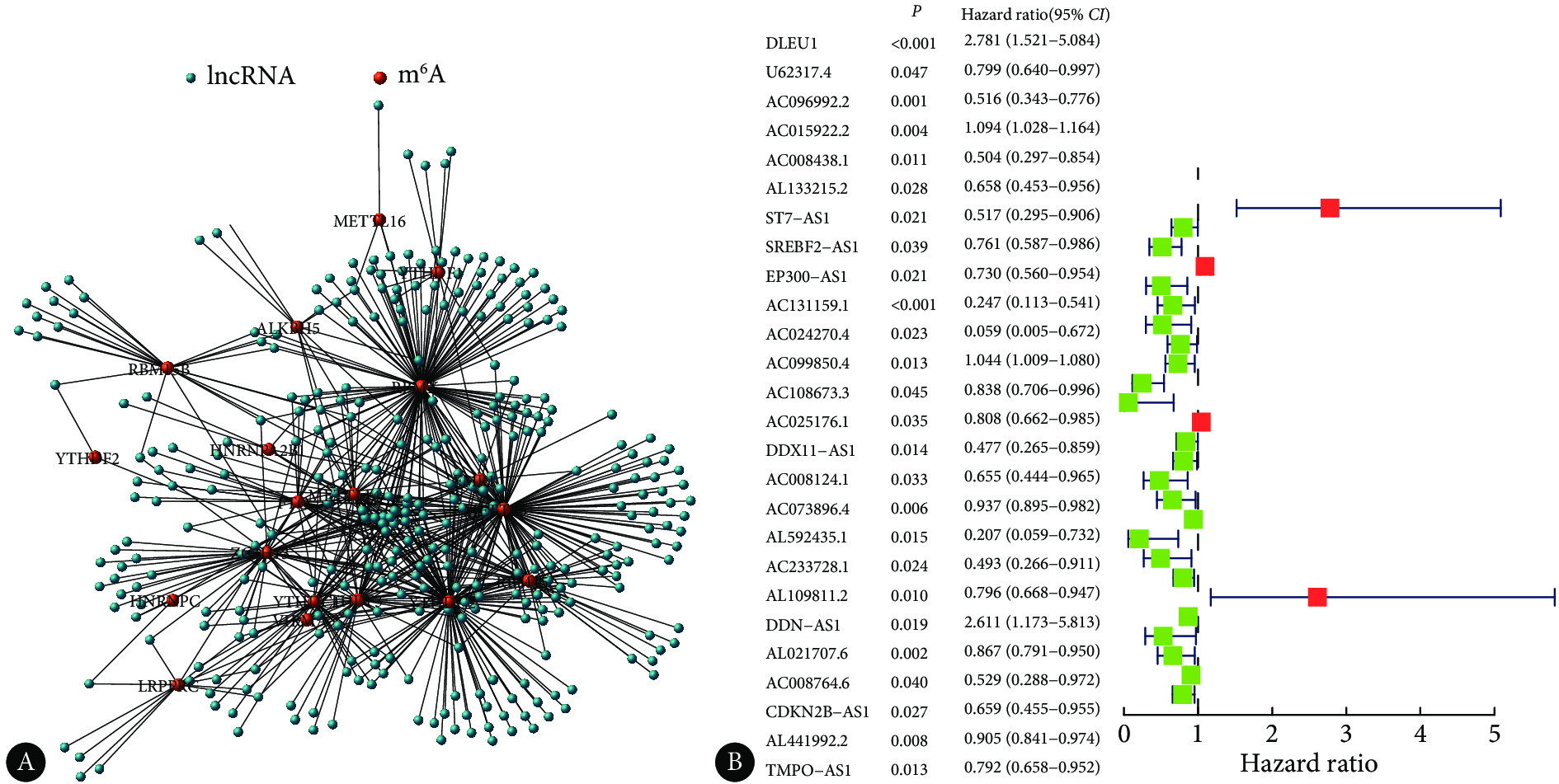
我们从TCGA数据库中下载309例宫颈癌患者的转录组数据及其病例资料,剔除5例临床数据不全的样本,最终共纳入304例。首先利用perl程序对数据进行整理和注释,并提取14086个lncRNA在宫颈癌患者样本中每千个碱基的转录每百万映射读取的碎片数(Fragments per Kilobase Million, FPKM)。我们挑选了与肿瘤密切相关的20个m6A调控因子,包括甲基转移酶(METTL3、METTL14、METTL16、WTAP、VIRMA、ZC3H13、RBM15、RBM15B),结合蛋白(YTHDC1、YTHDC2、YTHDF1、YTHDF2、YTHDF3、HNRNPC、FMR1、LRPPRC、HNRNPA2B1、RBMX),去甲基化酶(FTO、ALKBH5),利用R语言4.1.2中的“limma”软件包提取上述基因的表达矩阵,根据相关系数绝对值>0.3、P<0.001的滤过条件整合lncRNA表达谱,得到343个m6A修饰相关lncRNA,包括m6A调控因子具有相关关系的lncRNA、相关系数、调控关系及P 值。利用“limma”软件包提取宫颈癌患者的生存数据,其中生存时间以年为单位,生存状态以“0”和“1”分别表示生存和死亡,整合生存数据和343个m6A修饰相关lncRNA表达量,并对上述lncRNA表达量单个因素进行Cox回归分析该表达量是否与宫颈癌患者的预后相关,随后输出P<0.05具有预后价值的26个m6A修饰相关lncRNA,及其风险率(HR)、95%置信区间(CI)和P值。
1.2 聚类分析
按照指定聚类数目(k=2~9)从304例宫颈癌患者样本中抽取一定样本的数据集,利用“consensus cluster plus”软件包评估不同聚类数目下分组合理性,当累积分布函数(CDF)曲线达到近似最大值时,对应k值为最佳稳定性,结果显示k=2时组内差异最小,组间差异最大,聚类稳定性最佳,因此将患者分为cluster1(聚类1)和cluster2(聚类2)。利用“Survival”软件包绘制KM曲线比较两个组间OS是否有差异;据研究表明,宫颈癌患者采用免疫检查点(CTLA-4、PD-L1、PD-1)抑制剂的免疫治疗疗效甚佳 [14]。为了探究m6A修饰相关lncRNA与免疫检查点相关蛋白CTLA-4、PD-L1、PD-1的关系,我们通过“ggplot2”“ggpubr” “corrplot”软件包绘制PD-1、PD-L1、CTLA-4在两个亚组之间的差异表达箱形图,并进行PD-1、PD-L1、CTLA-4与m6A修饰相关lncRNA的相关性分析。从TIDE数据库中输入TCGA宫颈癌样本数据后计算样本TIDE评分,利用“ggpubr” “limma”软件包分析高低风险组间的TIDE差异。免疫浸润是肿瘤躲避免疫系统监视过程中的关键,能够改变患者的临床结局[15],因此了解宫颈癌的免疫浸润有助于提高其免疫治疗反应率,为制定新的免疫治疗策略提供更多的理论依据,我们从CIBERSORT网站(http://CIBERSORT.stanford.edu/)下载22个免疫细胞亚群的LM22基因集文件,通过对宫颈癌样本的表达矩阵进行非负矩阵分解从而计算不同免疫细胞所占比例,分析免疫细胞在宫颈癌样本中的浸润情况,并使用“corplot”“vioplot”和“ggplot2”软件包使结果可视化。
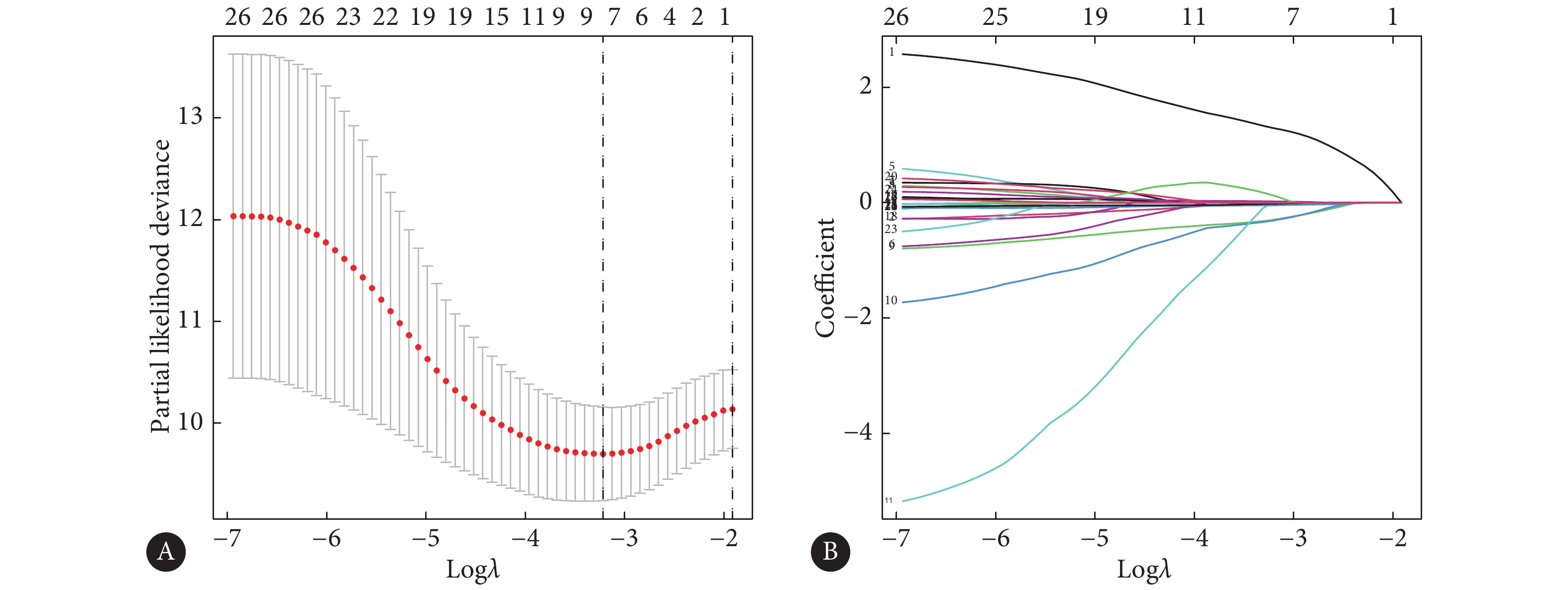
1.3 预后风险模型构建及评估
通过“Survival”“glmnet”“caret”软件包对通过单因素Cox分析得到的与宫颈癌预后相关的m6A修饰相关lncRNA进行Lasso回归分析,构建cvfit和lambda两个内置函数优化模型用以变量取值。根据函数“λ”值来控制变量个数,“λ”越大对变量较多的线性模型过拟合程度就越大,结果显示“λ”取值为7时模型拟合程度最佳,由此我们鉴定出DLEU1、EP300-AS1、AC131159.1、AC099850.4、DDN-AS1、AL441992.2、AL021707.6用于构建预后风险模型。我们对基因的相关回归系数进行加权计算每个宫颈癌患者样本的风险评分,即风险评分=(DLEU1表达量×1.30005246896042)+(EP300-AS1表达量×−0.299555310011884)+(AC131159.1表达量×−0.311617140344044)+(AC099850.4表达量×0.023992922627114)+(DDN-AS1表达量×0.133059169788479)+(AL021707.6表达量×−0.0435711218239825)+(AL441992.2表达量×−0.0175865350548823)。我们采用交叉验证法中的5∶5留出法对样本进行随机抽样,即随机抽取样本的50%用来做训练集,其余50%做测试集,两数据集互斥且样本分布基本一致。通过对训练集样本进行风险评分得到其中位值,根据该值将患者分为高、低风险组,根据训练集的风险评分中位值将测试集中的患者也分为高、低风险组。为了验证高、低风险组的划分是否合理,我们通过PCA分析函数对宫颈癌样本中所有基因表达谱、20个m6A调控因子表达量、343个m6A修饰相关lncRNA表达量和用以构建风险模型的7个lncRNA表达量通过正交交换进行降维、模型识别和分组可视化。我们利用“pheatmap”“survival”“timeROC”“survminer”软件包在训练集和测试集中比较高、低风险组间OS、风险趋势、ROC 曲线下面积(AUC)差异和临床分组的模型验证。最后通过“ggplot2”“ggpubr”“ggExtra”软件包对宫颈癌样本的风险评分与22种免疫细胞进行相关性检验,并将结果可视化。
1.4 统计学方法
采用双侧log-rank检验和Kaplan-Meier分析评估组间OS,利用Pearson相关性检验鉴定具有宫颈癌预后价值的m6A修饰相关lncRNA,并分析PD1和PD-L1差异表达和免疫浸润水平。采用单因素和多因素Cox回归分析,以验证宫颈癌的独立预后因素,并计算HR和95%CI。采用Wilcoxon检验分析两组变量之间的差异。双侧P<0.05为差异有统计学意义,所有统计分析均采用R语言Rx64 4.1.2。
2. 结果
2.1 一般资料及m6A修饰相关lncRNA在TCGA宫颈癌数据库中的表达情况
本研究共纳入304例宫颈癌患者,包括3例正常组织样本和301例肿瘤组织样本,平均年龄为48岁,均为女性,存活233例,死亡71例(表1)。
表 1 304例宫颈癌患者的一般病理资料Table 1. The clinicopathologic information of 304 patients with cervical cancerVariable Classification Case (%) Age ≤50 yr. 117 (38.49) >50 yr. 187 (61.51) Survival states Alive 233 (76.64) Death 71 (23.36) Pathological grades G1 18 (5.92) G2 136 (44.74) G3 120 (39.47) G4 1 (0.33) Gx 29 (9.54) Clinical stages ⅠB 57 (18.75) ⅡA 14 (4.61) ⅡB 7 (2.30) ⅢA 1 (0.33) ⅢB 25 (8.22) ⅣA 3 (0.99) ⅣB 10 (3.29) Unknown 187 (61.51) T Tis 1 (0.33) T1 141 (46.38) T2 72 (23.68) T3 21 (6.91) T4 10 (3.29) Tx 59 (19.41) M M0 116 (38.16) M1 10 (3.29) Mx 178 (58.55) N N0 135 (44.41) N1 60 (19.74) Nx 109 (35.86) 我们提取20个m6A调控因子和14086个lncRNA的FPKM值,筛选出343个m6A修饰相关lncRNA(图1A)。利用单因素Cox分析得到26个m6A修饰相关lncRNA与宫颈癌患者预后相关,其中DLEU1、AC015922.2、AC099850.4、DDN-AS1与宫颈癌患者的OS呈负相关,而其余22个m6A修饰相关lncRNA与宫颈癌患者的OS呈正相关(图1B)。
2.2 聚类分析具有预后价值的m6A修饰相关lncRNA
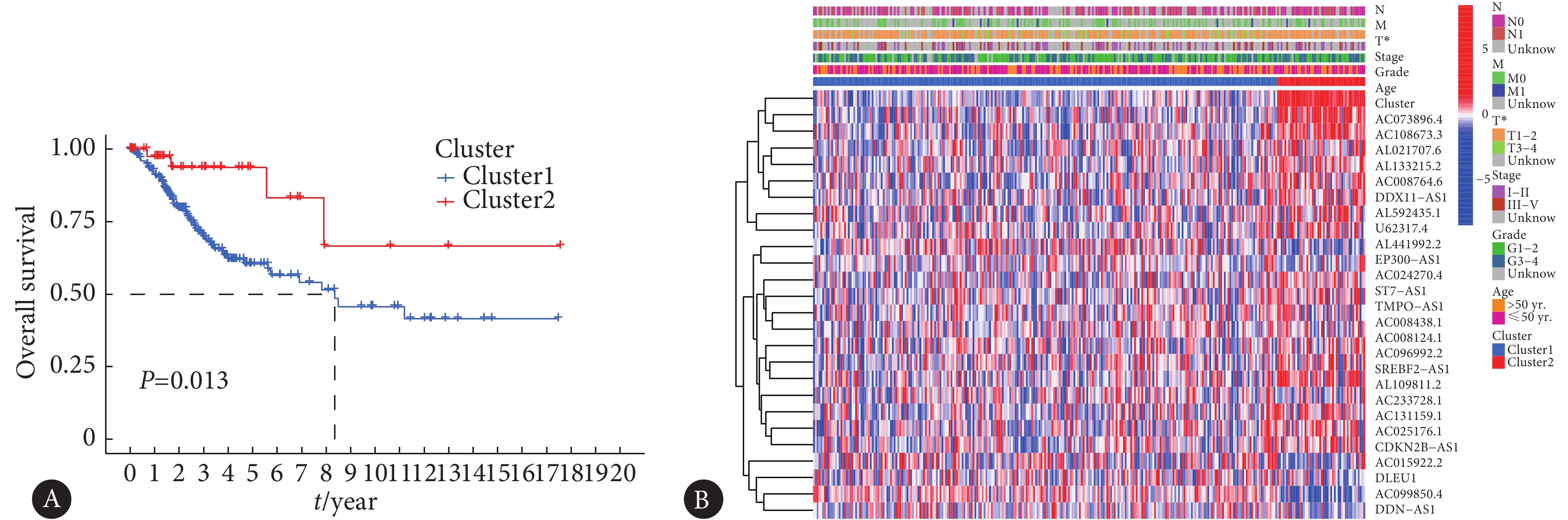
为了探究上述26个m6A修饰相关lncRNA在宫颈癌预后中的作用,我们采用聚类分析将304例宫颈癌样本分为cluster1和cluster2两组。Kaplan-Meier曲线显示cluster1组的患者比cluster2组的预后更差(图2A)。聚类热图表明临床T分期较高的患者集中分布于cluster1组,但在年龄等变量上两组并无差别(图2B)。
![]() 图 2 聚类分析具有宫颈癌预后意义的m6A修饰相关lncRNAFigure 2. Cluster analysis of m6A modification-related lncRNAs with prognostic value in cervical cancer patientsA: The Kaplan-Meier curves compared the OS of cluster1 and cluster2; B: The relation heatmap of lncRNAs related with m6A modification in cluster1 and cluster2 group, *P<0.05.
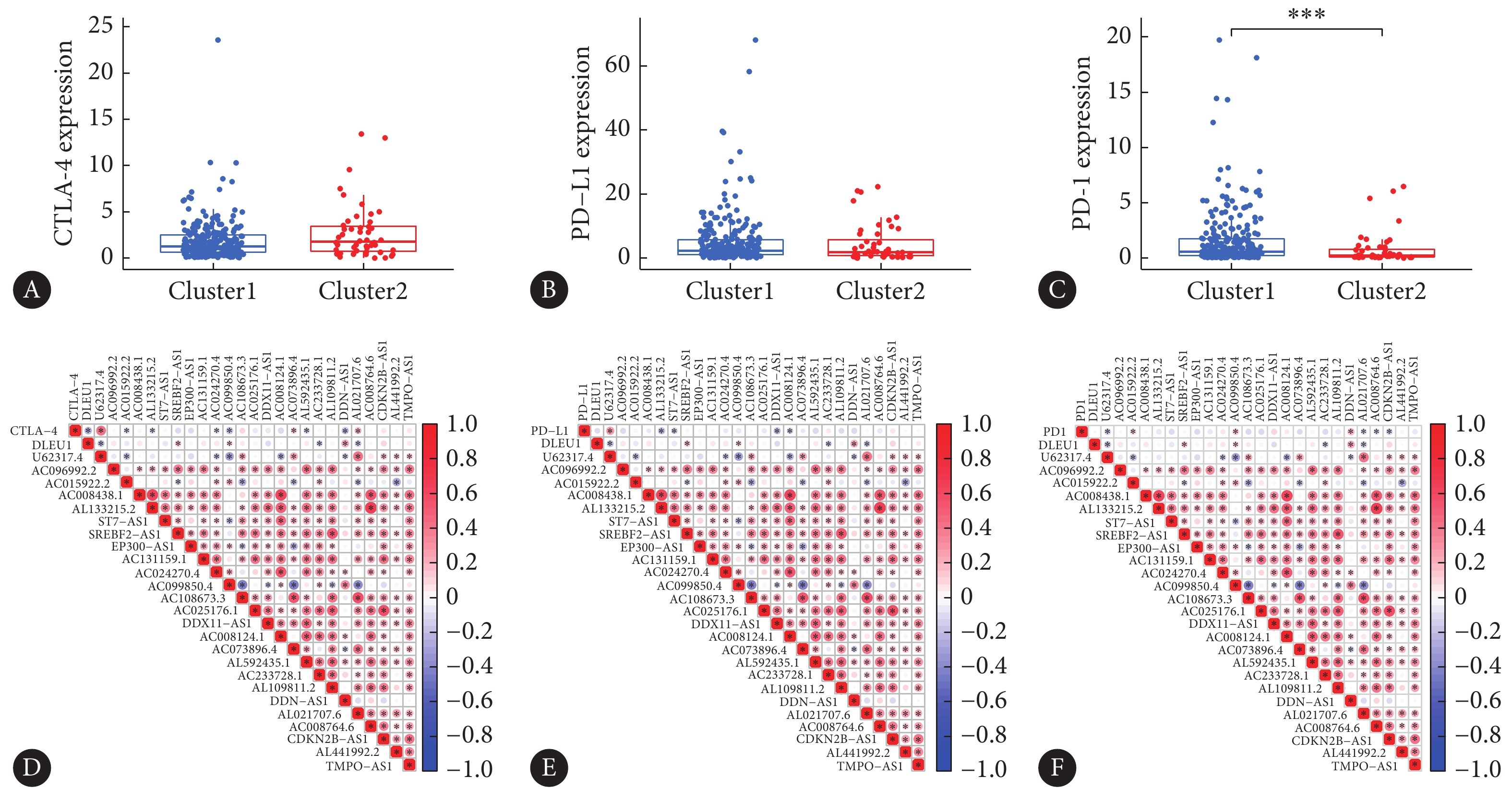
图 2 聚类分析具有宫颈癌预后意义的m6A修饰相关lncRNAFigure 2. Cluster analysis of m6A modification-related lncRNAs with prognostic value in cervical cancer patientsA: The Kaplan-Meier curves compared the OS of cluster1 and cluster2; B: The relation heatmap of lncRNAs related with m6A modification in cluster1 and cluster2 group, *P<0.05.我们通过比较CTLA-4、PD-L1、 PD-1在两种聚类亚组中的表达水平发现,PD-L1与CTLA-4的表达在两组间差异无统计学意义(图3A、3B),而PD-1在cluster1组中的表达明显高于cluster2组(图3C)。CTLA-4、PD-L1、PD-1与上述lncRNA之间的相关性热图结果显示CTLA-4的表达与DLEU1、AC015922.2、AL133215.2、AC024270.4、AC099850.4、AC073896.4、AL592435.1、DDN-AS1、AL021707.6以及U62317.4表达相关(图3D); PD-L1与AL133215.2、ST7-AS1、DDX11-AS1、AC008124.1以及U62317.4的表达相关(图3E);PD-1与U62317.4、AC015922.2、AC099850.4、SREBF2-AS1、DDN-AS1、AL021707.6、AC008764.6以及CDKN2B-AS1表达相关(图3F)。
![]() 图 3 CTLA-4(A)、PD-L1(B)、PD-1(C)在cluster1/2亚组中的差异表达及CTLA-4(D)、PD-L1(E)、PD-1(F)与m6A修饰相关lncRNA之间的相关性Figure 3. The differential expression of CTLA-4 (A) , PD-L1 (B) , and PD-1 (C) in the cluster1/2 group and the correlation between CTLA-4 (D), PD-L1 (E), and PD-1 (F) and lncRNAs related with m6A modification*P<0.05, ***P<0.001.
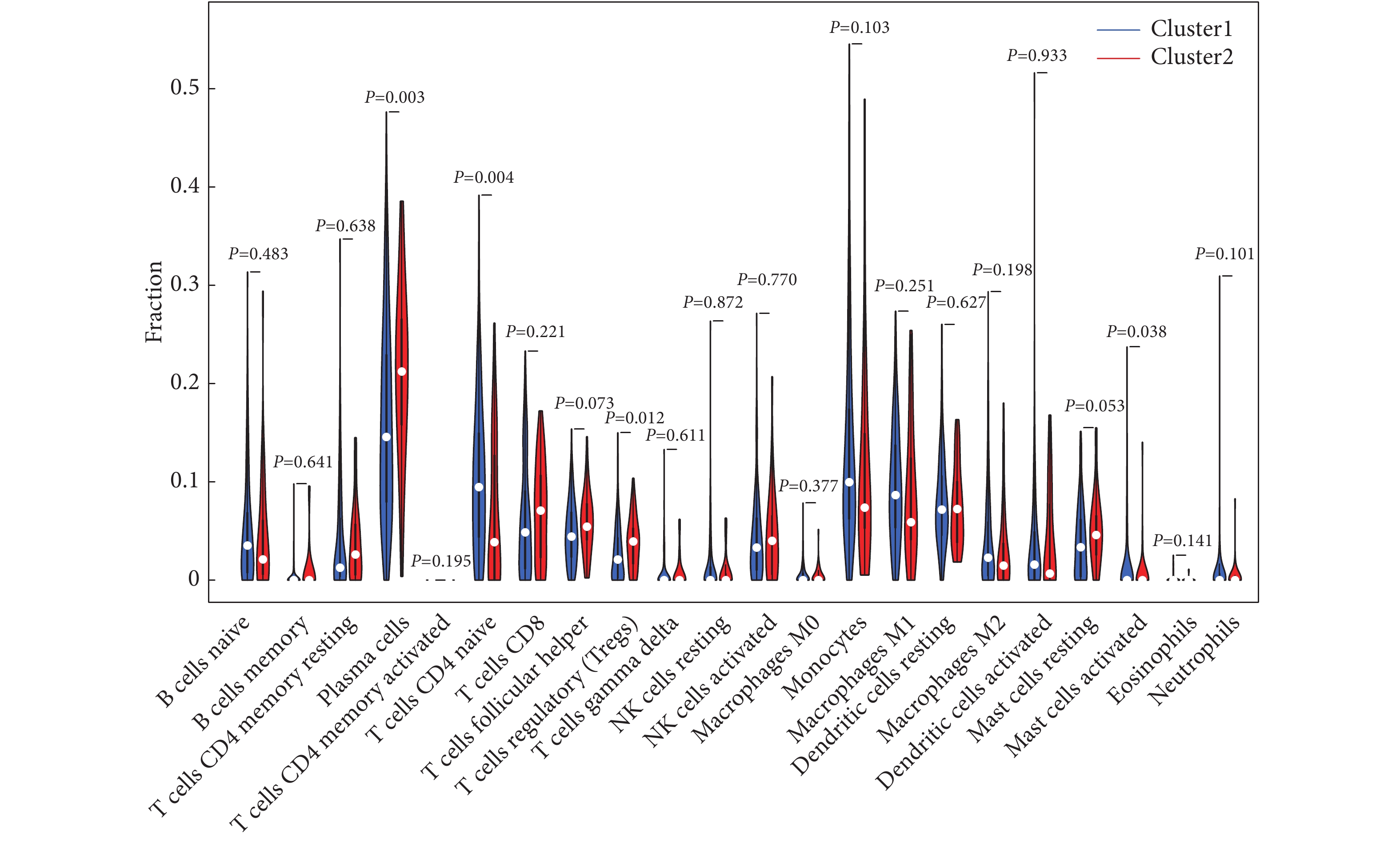
图 3 CTLA-4(A)、PD-L1(B)、PD-1(C)在cluster1/2亚组中的差异表达及CTLA-4(D)、PD-L1(E)、PD-1(F)与m6A修饰相关lncRNA之间的相关性Figure 3. The differential expression of CTLA-4 (A) , PD-L1 (B) , and PD-1 (C) in the cluster1/2 group and the correlation between CTLA-4 (D), PD-L1 (E), and PD-1 (F) and lncRNAs related with m6A modification*P<0.05, ***P<0.001.通过比较cluster1和cluster2两组中免疫浸润细胞的组成差异,发现cluster1组CD4阳性记忆T细胞的浸润更多,cluster2组CD8阳性T细胞、调节性T细胞以及滤泡辅助T细胞的浸润更多(图4)。
2.3 m6A修饰相关lncRNA构建预后风险模型及验证
为了评估m6A修饰相关lncRNA在宫颈癌预后中的预测价值,我们利用Lasso回归分析从上述26个m6A修饰相关lncRNA中鉴定出7个lncRNA用于构建预后风险模型,即DLEU1、EP300-AS1、AC131159.1、AC099850.4、DDN-AS1、AL441992.2和AL021707.6(图5)。同时,我们根据留出法将304例宫颈癌样本分成训练集和测试集,用以验证该模型的预测能力。
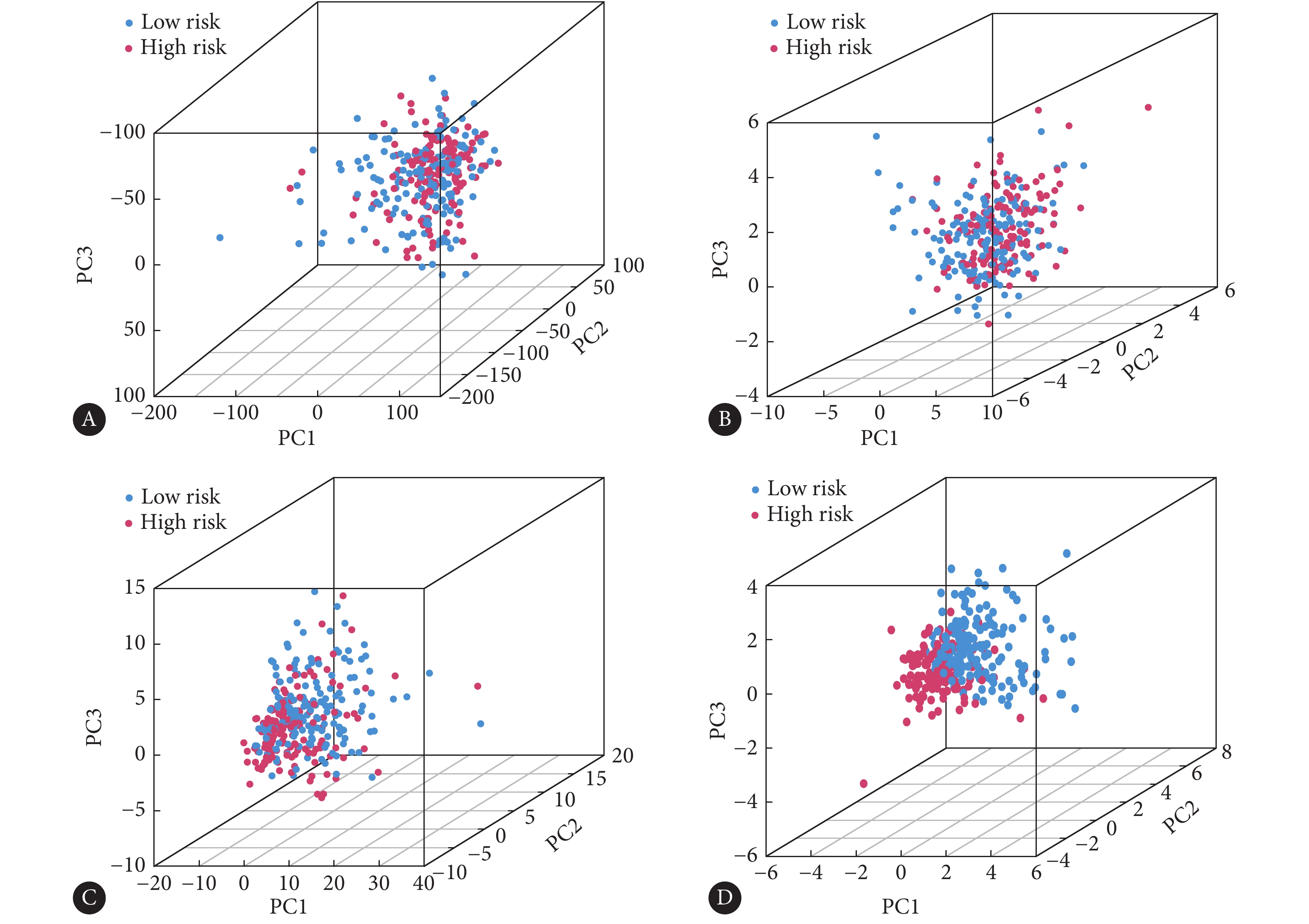
根据训练集风险评分中位值将训练集和测试集中样本分别划分成高风险组和低风险组。为了评估预后特征对高、低风险组的划分是否合理,我们通过PCA主成分发现宫颈癌样本中所有基因表达谱、20个m6A调控因子表达量、343个m6A修饰相关lncRNA表达量的高、低风险组分布相对离散(图6A~6C),而基于我们的预后风险模型所划分的高、低风险组组内分布集中,组间区分明显(图6D),说明预后风险模型能够更加合理有效地对高、低风险组进行划分。
![]() 图 6 TCGA宫颈癌数据库转录组全基因(A)、m6A修饰调控因子(B)、m6A修饰相关lncRNA(C)、构建预后风险模型的m6A修饰相关lncRNA(D)的PCA主成分分析Figure 6. PCA principal component analysis of the transcriptome of cervical cancer from TCGA database (A), m6A regulatory factors (B), lncRNA associated with m6A modification (C), lncRNA associated with m6A modification used to construct prognostic risk models (D)
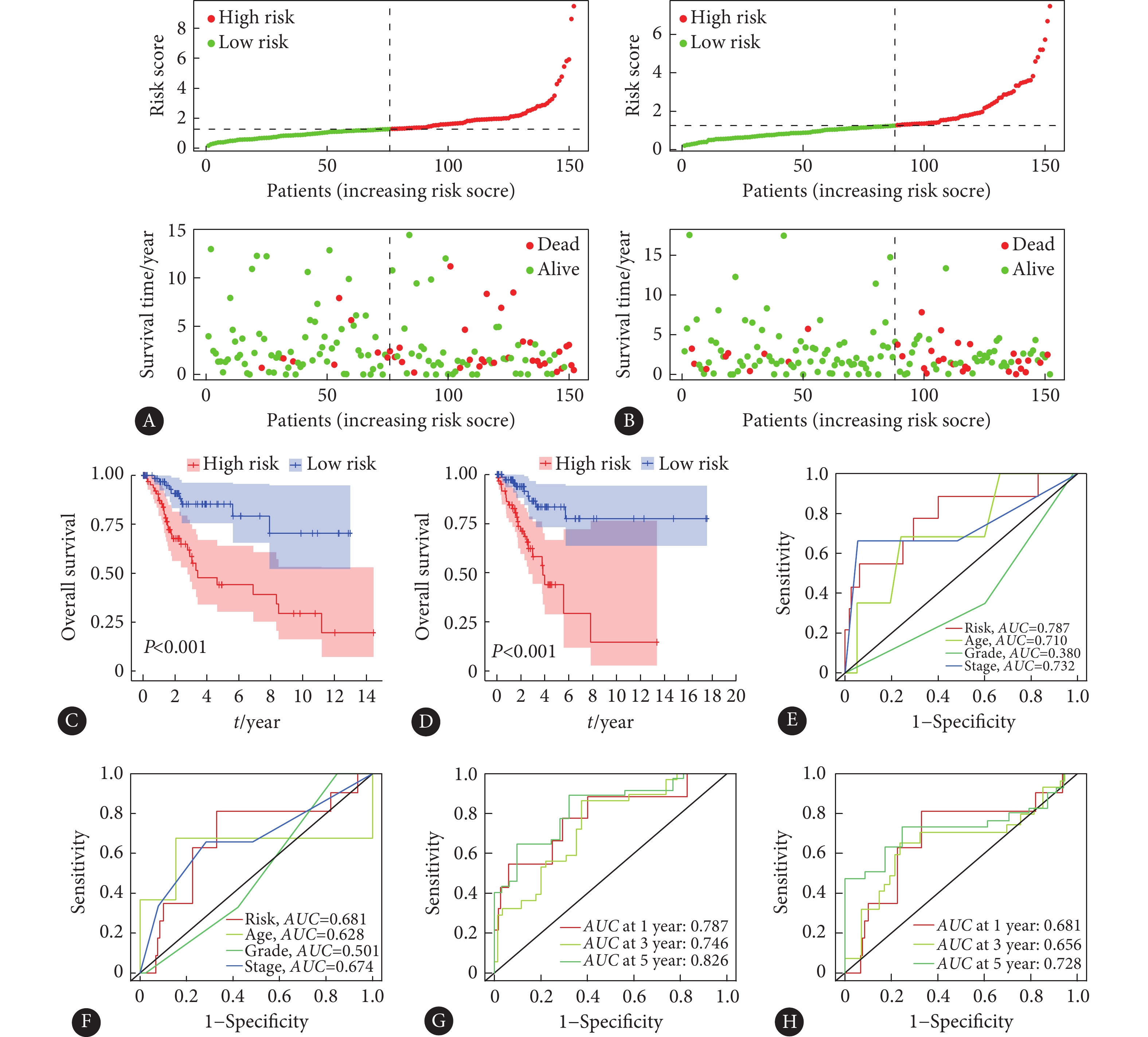
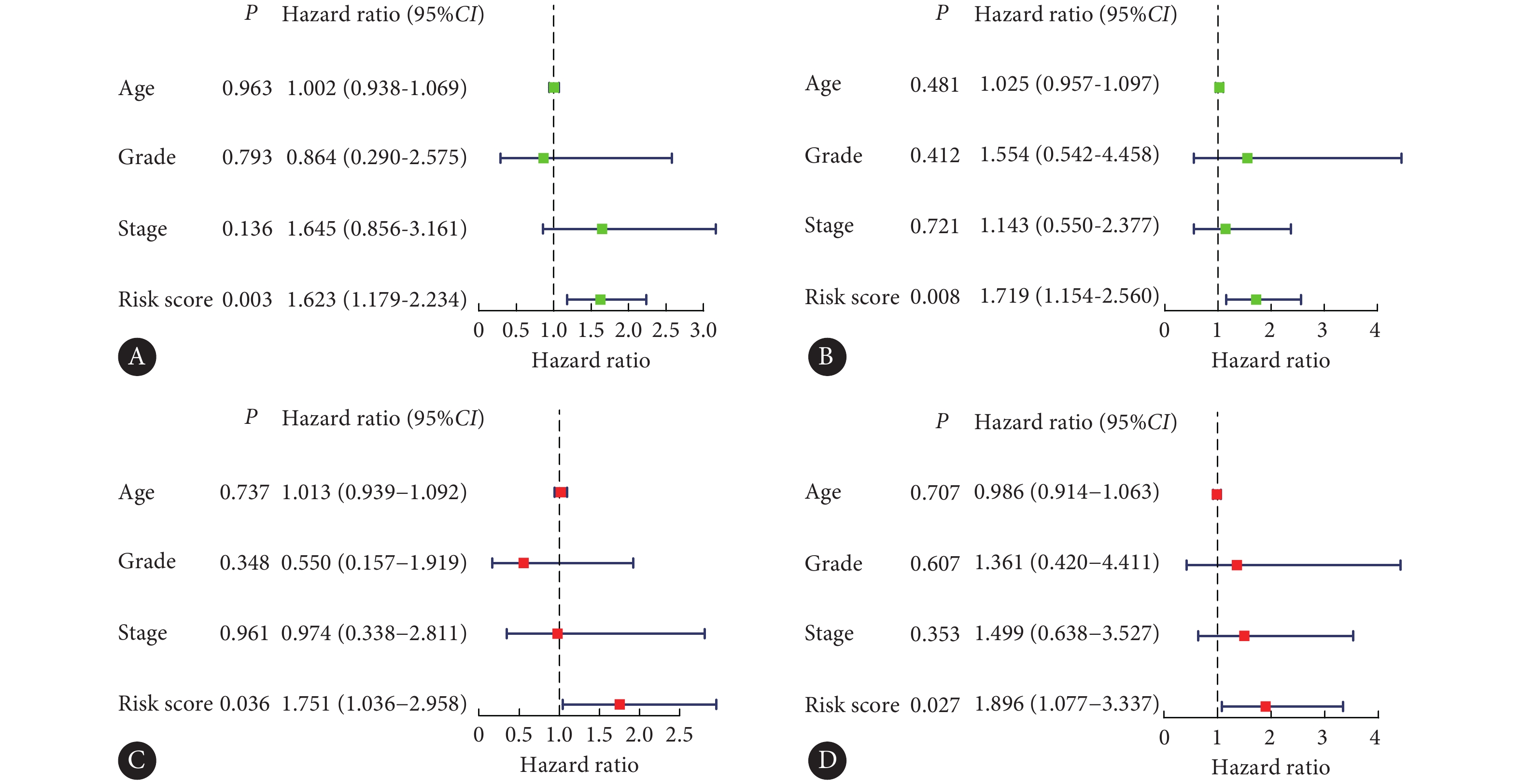
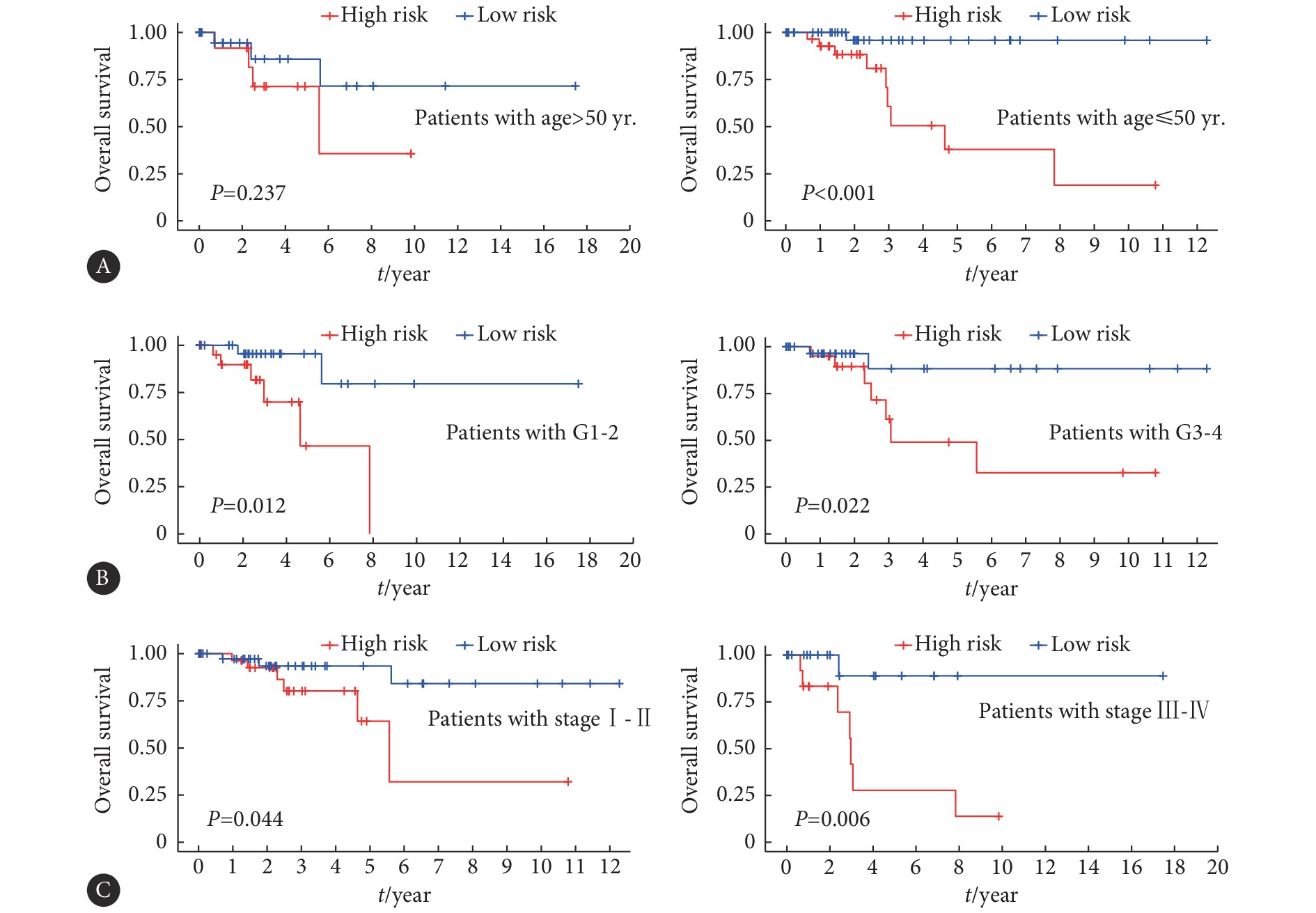
图 6 TCGA宫颈癌数据库转录组全基因(A)、m6A修饰调控因子(B)、m6A修饰相关lncRNA(C)、构建预后风险模型的m6A修饰相关lncRNA(D)的PCA主成分分析Figure 6. PCA principal component analysis of the transcriptome of cervical cancer from TCGA database (A), m6A regulatory factors (B), lncRNA associated with m6A modification (C), lncRNA associated with m6A modification used to construct prognostic risk models (D)我们绘制了训练集和测试集风险趋势曲线(图7A、7B)和KM曲线(图7C、7D),两数据集的风险趋势曲线均表明随着患者风险指数的增加,生存率逐渐降低,KM曲线显示高风险组OS均远远低于低风险组的OS。为了明确该预后风险模型对评估宫颈癌患者预后的准确性,我们以两数据集的风险评分、年龄、病理学分级、临床分期(图7E、7F),生存期1年、3年、5年(图7G、7H)指标作为连续变量绘制ROC曲线。通过对AUC(取值范围为0.5~1.0)评估,结果显示该预后风险模型评估5年OS的准确性最佳,对于风险评分等指标也具有较好的评估意义。我们通过单因素Cox分析分别评估年龄、病理学分级、临床TMN分期、风险评分在训练集和测试集中对宫颈癌预后的预测价值,分析表明仅风险评分具有显著的预后预测价值(图8A、8B)。多因素Cox回归分析上述变量在训练集和测试集中对宫颈癌预后的独立预测价值,结果显示只有风险评分作为独立预后因素与OS相关(图8C、8D)。我们对高、低风险组的临床病理特征进行分层分析OS差异,结果表明年龄、病理学分级、临床TMN分期亚组中高风险组OS均低于低风险组(图9)。
![]() 图 7 预后风险模型的验证Figure 7. Validation of the prognostic risk modelThe classification of high risk and low risk groups in the training cohort (A) and the validation cohort (B), with the dotted line to the left representing the low-risk groups and the dotted line to the right representing the high risk groups (top graphs); the distribution diagram of survival time and state in two groups (bottom graphs). Kaplan-Meier curves of high risk and low risk groups in training cohort (C) and validation cohort (D). ROC curves of risk score, age, pathology grade, and clinical stage variables in training cohort (E) and validation cohort (F). ROC curves of 1-year, 3-year, and 5-year survival prediction in training cohort (G) and validation cohort (H).
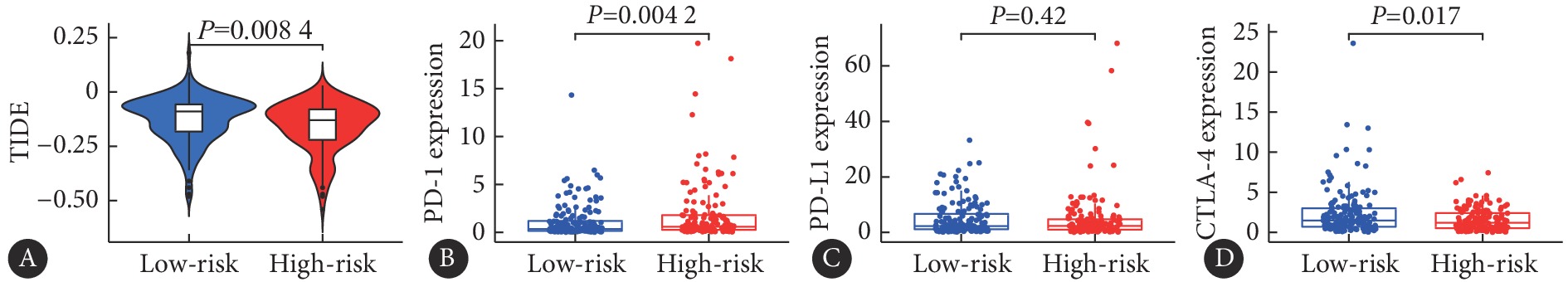
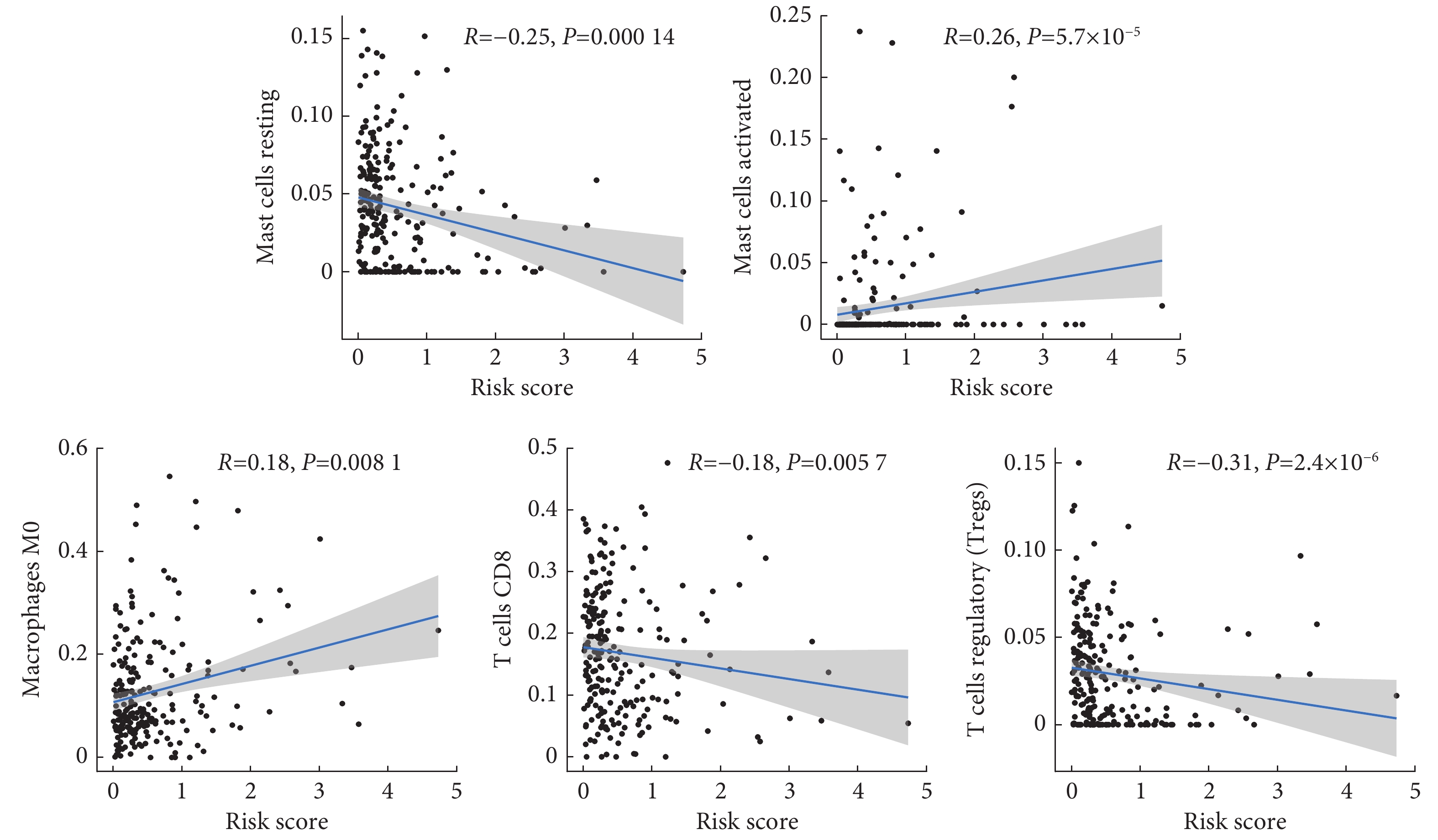
图 7 预后风险模型的验证Figure 7. Validation of the prognostic risk modelThe classification of high risk and low risk groups in the training cohort (A) and the validation cohort (B), with the dotted line to the left representing the low-risk groups and the dotted line to the right representing the high risk groups (top graphs); the distribution diagram of survival time and state in two groups (bottom graphs). Kaplan-Meier curves of high risk and low risk groups in training cohort (C) and validation cohort (D). ROC curves of risk score, age, pathology grade, and clinical stage variables in training cohort (E) and validation cohort (F). ROC curves of 1-year, 3-year, and 5-year survival prediction in training cohort (G) and validation cohort (H).为了评估高低风险组患者接受免疫检查点抑制剂(ICI)治疗后的预后效果,我们采用TIDE评分分析发现高风险组的TIDE评分明显低于低风险组,说明低风险组TIDE评分越高,发生免疫逃避的可能性越大,患者接受ICI治疗后预后效果越差(图10A)。同时在预后风险模型中验证PD-L1、PD-1、CTLA-4 ICI的可行性,发现PD-1在高风险组中表达量更高(图10B),PD-L1在两组间的表达没有差异(图10C),而CTLA-4在低风险组中表达较高(图10D)。我们进一步对风险评分与22种免疫细胞的相关性进行评估,发现风险评分与M0型巨噬细胞(P<0.001)、活化型巨大细胞(P<0.001)存在正相关,而与静止型巨大细胞(P<0.001)、CD8阳性的T细胞(P<0.001)、调节T细胞(P<0.001)存在负相关(图11)。
3. 讨论
lncRNA不仅能够通过参与多种肿瘤相关信号通路如p53[5]、NF-κB[16]、PI3K/AKT[17]和Notch[18]等信号通路,调控原癌基因或抑癌基因的表达,而且能作为评估肿瘤患者预后的重要生物标志物[19]。随着对lncRNA功能研究逐渐深入,研究人员发现lncRNA能够作为评估肿瘤组织免疫浸润及免疫治疗的重要标志物。例如在非小细胞肺癌中,与免疫浸润相关lncRNA高表达的患者其预后明显较差[20]。HLA-DQB1-AS1等lncRNA能够作为黑色素瘤预后标志物评估免疫浸润的程度,和预测免疫治疗改善黑色素瘤患者OS的指标[21]。近年来,lncRNA的表观遗传调控逐渐引起重视,m6A甲基化修饰与肿瘤的转移、耐药、免疫应答具有显著的相关性[22]。据此,我们针对m6A修饰相关lncRNA在宫颈癌预后预测中的价值进行深入的挖掘和分析,从而筛选出作为宫颈癌预后及免疫治疗疗效预测的生物标志物。
本研究中我们通过Lasso回归分析以7个m6A修饰相关lncRNA联合预后标志物,即DLEU1、EP300-AS1、AC131159.1、AC099850.4、DDN-AS1、AL441992.2、AL021707.6构建宫颈癌的预后风险模型。已有研究证明,DLEU1是预测宫颈癌预后的lncRNA之一,且其表达与宫颈癌患者的预后呈负相关[23,24]。EP300-AS1在宫颈癌的样本组织中存在基因突变,该突变可能是导致宫颈癌预后不良的重要因素之一[25]。在宫颈癌中高表达的DDN-AS1也已被证实与宫颈癌的预后不良存在密切关系,能通过DDN-AS1-miR-15a/16-TCF3反馈环促进宫颈癌的恶化[26]。 AL021707.6被证明与细胞凋亡的进程高度相关[27]。研究人员曾以单个lncRNA及其涉及通路作为预测宫颈癌预后的标志物进行分析[28]。例如,ZENG等[29]发现,lncRNA SNHG7在宫颈癌组织中呈高表达,从而促进宫颈癌的增殖和转移,同时其表达增高导致宫颈癌患者的OS明显降低。然而ZHANG等[30]的研究表明,多个lncRNA所构建的预后风险模型要明显优于单个lncRNA预测的灵敏度和特异性。因此,本研究所得到的7个m6A修饰相关lncRNA联合预测宫颈癌预后的风险模型具有更高的应用价值。
已有研究证明,复发性、转移性的宫颈癌患者在接受纳武利尤单抗(PD1单克隆抗体)治疗后中位生存期延长,客观缓解率达到26.3%[31]。且FDA已批准PD-1单克隆抗体——帕博利珠单抗作为化疗后进展的晚期宫颈癌患者的抗PD-1疗法[32]。在本研究中,TIDE评分预测高风险组接受ICI疗法后预后更佳,且PD-1在预后较差的cluster1组以及高风险组中表达较高,该两组中CD8阳性T细胞的浸润明显较多。有研究指出PD-1协同CD8阳性T细胞浸润能够提高免疫应答的反应率[33],说明高风险组接受PD-1治疗后能获益更大,充分显示PD-1作为宫颈癌的免疫治疗靶点具备可行性。此外,在预后风险模型中AC099850.4、DDN-AS1、AL021707.6与PD-1的表达具有相关性,说明我们所建立的预后风险模型能提供评估宫颈癌以PD-1作为免疫治疗靶点的预测指标。
本研究也存在一些不足。首先,此研究是基于TCGA数据库进行预测宫颈癌预后的模型分析,仅采用了生物信息学的方法,预后风险模型及结果都需要进一步的分子实验来验证。其次,本次研究样本量较少,后续应当扩大数据库的样本量及多中心收集宫颈癌临床样本进行验证。尽管存在这些不足之处,但本研究阐明了m6A修饰相关lncRNA在宫颈癌预后中的预测价值,为PD-1免疫治疗宫颈癌的疗效评估提供了预测指标。
综上,通过单因素、多因素Cox、Lasso回归等分析鉴定出DLEU1、EP300-AS1、AC131159.1、AC099850.4、DDN-AS1、AL441992.2、AL021707.6这7个m6A修饰相关lncRNA建立的预后风险模型对宫颈癌患者的预后具有独立的预测意义,同时该模型能够在治疗前对PD-1作为免疫治疗靶点的可行性和疗效进行评估。这些发现更有助于我们了解m6A修饰相关lncRNA在宫颈癌预后中的价值,为宫颈癌免疫治疗策略的制定提供重要的生物学指标和理论依据。
* * *
利益冲突 所有作者均声明不存在利益冲突
-
图 2 聚类分析具有宫颈癌预后意义的m6A修饰相关lncRNA
Figure 2. Cluster analysis of m6A modification-related lncRNAs with prognostic value in cervical cancer patients
A: The Kaplan-Meier curves compared the OS of cluster1 and cluster2; B: The relation heatmap of lncRNAs related with m6A modification in cluster1 and cluster2 group, *P<0.05.
图 3 CTLA-4(A)、PD-L1(B)、PD-1(C)在cluster1/2亚组中的差异表达及CTLA-4(D)、PD-L1(E)、PD-1(F)与m6A修饰相关lncRNA之间的相关性
Figure 3. The differential expression of CTLA-4 (A) , PD-L1 (B) , and PD-1 (C) in the cluster1/2 group and the correlation between CTLA-4 (D), PD-L1 (E), and PD-1 (F) and lncRNAs related with m6A modification
*P<0.05, ***P<0.001.
图 6 TCGA宫颈癌数据库转录组全基因(A)、m6A修饰调控因子(B)、m6A修饰相关lncRNA(C)、构建预后风险模型的m6A修饰相关lncRNA(D)的PCA主成分分析
Figure 6. PCA principal component analysis of the transcriptome of cervical cancer from TCGA database (A), m6A regulatory factors (B), lncRNA associated with m6A modification (C), lncRNA associated with m6A modification used to construct prognostic risk models (D)
图 7 预后风险模型的验证
Figure 7. Validation of the prognostic risk model
The classification of high risk and low risk groups in the training cohort (A) and the validation cohort (B), with the dotted line to the left representing the low-risk groups and the dotted line to the right representing the high risk groups (top graphs); the distribution diagram of survival time and state in two groups (bottom graphs). Kaplan-Meier curves of high risk and low risk groups in training cohort (C) and validation cohort (D). ROC curves of risk score, age, pathology grade, and clinical stage variables in training cohort (E) and validation cohort (F). ROC curves of 1-year, 3-year, and 5-year survival prediction in training cohort (G) and validation cohort (H).
表 1 304例宫颈癌患者的一般病理资料
Table 1 The clinicopathologic information of 304 patients with cervical cancer
Variable Classification Case (%) Age ≤50 yr. 117 (38.49) >50 yr. 187 (61.51) Survival states Alive 233 (76.64) Death 71 (23.36) Pathological grades G1 18 (5.92) G2 136 (44.74) G3 120 (39.47) G4 1 (0.33) Gx 29 (9.54) Clinical stages ⅠB 57 (18.75) ⅡA 14 (4.61) ⅡB 7 (2.30) ⅢA 1 (0.33) ⅢB 25 (8.22) ⅣA 3 (0.99) ⅣB 10 (3.29) Unknown 187 (61.51) T Tis 1 (0.33) T1 141 (46.38) T2 72 (23.68) T3 21 (6.91) T4 10 (3.29) Tx 59 (19.41) M M0 116 (38.16) M1 10 (3.29) Mx 178 (58.55) N N0 135 (44.41) N1 60 (19.74) Nx 109 (35.86) -
[1] ARBYN M, WEIDERPASS E, BRUNI L, et al. Estimates of incidence and mortality of cervical cancer in 2018: A worldwide analysis. Lancet Glob Health, 2020, 8 (2): e191-e203[2022-01-14]. https://doi.org/10.1016/S2214-109X(19)30482-6.
[2] LIONTOS M, KYRIAZOGLOU A, DIMITRIADIS I, et al. Systemic therapy in cervical cancer: 30 years in review. Crit Rev Oncol Hematol,2019,137: 9–17. DOI: 10.1016/j.critrevonc.2019.02.009
[3] JOHNSON C A, JAMES D, MARZAN A, et al. Cervical cancer: An overview of pathophysiology and management. Semin Oncol Nurs,2019,35(2): 166–174. DOI: 10.1016/j.soncn.2019.02.003
[4] BILLAN S, KAIDAR-PERSON O, GIL Z. Treatment after progression in the era of immunotherapy. Lancet Oncol, 2020, 21 (10): e463−e476[2022-01-14]. https://doi.org/10.1016/S1470-2045(20)30328-4.
[5] WEI G H, WANG X. lncRNA MEG3 inhibit proliferation and metastasis of gastric cancer via p53 signaling pathway. Eur Rev Med Pharmacol Sci,2017,21(17): 3850–3856.
[6] REN J, DING L, ZHANG D, et al. Carcinoma-associated fibroblasts promote the stemness and chemoresistance of colorectal cancer by transferring exosomal lncRNA H19. Theranostics,2018,8(14): 3932–3948. DOI: 10.7150/thno.25541
[7] DAI D, WANG H, ZHU L, et al. N6-methyladenosine links RNA metabolism to cancer progression. Cell Death Dis,2018,9(2): 124. DOI: 10.1038/s41419-017-0129-x
[8] ZHANG H, SHI X, HUANG T, et al. Dynamic landscape and evolution of m6A methylation in human. Nucleic Acids Res,2020,48(11): 6251–6264. DOI: 10.1093/nar/gkaa347
[9] PARK J W, HAN J W. Targeting epigenetics for cancer therapy. Arch Pharm Res,2019,42(2): 159–170. DOI: 10.1007/s12272-019-01126-z
[10] TU Z, WU L, WANG P, et al. N6-methylandenosine-related lncRNAs are potential biomarkers for predicting the overall survival of lower-grade glioma patients. Front Cell Dev Biol, 2020, 8: 642[2022-01-14]. https://doi.org/10.3389/fcell.2020.00642.
[11] MA S, CHEN C, JI X, et al. The interplay between m6A RNA methylation and noncoding RNA in cancer. J Hematol Oncol,2019,12(1): 121. DOI: 10.1186/s13045-019-0805-7
[12] WANG Y, LU J H, WU Q N, et al. LncRNA LINRIS stabilizes IGF2BP2 and promotes the aerobic glycolysis in colorectal cancer. Mol Cancer,2019,18(1): 174. DOI: 10.1186/s12943-019-1105-0
[13] WANG Q, GUO X, LI L, et al. N(6)-methyladenosine METTL3 promotes cervical cancer tumorigenesis and Warburg effect through YTHDF1/HK2 modification. Cell Death Dis,2020,11(10): 911. DOI: 10.1038/s41419-020-03071-y
[14] KAGABU M, NAGASAWA T, SATO C, et al. Immunotherapy for uterine cervical cancer using checkpoint inhibitors: Future directions. Int J Mol Sci,2020,21(7): 2335. DOI: 10.3390/ijms21072335
[15] ZHANG Y, ZHANG Z. The history and advances in cancer immunotherapy: Understanding the characteristics of tumor-infiltrating immune cells and their therapeutic implications. Cell Mol Immunol,2020,17(8): 807–821. DOI: 10.1038/s41423-020-0488-6
[16] PAN J X. LncRNA H19 promotes atherosclerosis by regulating MAPK and NF-kB signaling pathway. Eur Rev Med Pharmacol Sci,2017,21(2): 322–328.
[17] HUANG Y, ZHANG J, HOU L, et al. LncRNA AK023391 promotes tumorigenesis and invasion of gastric cancer through activation of the PI3K/Akt signaling pathway. J Exp Clin Cancer Res,2017,36(1): 194. DOI: 10.1186/s13046-017-0666-2
[18] WAN Y, YANG Z Q. LncRNA NEAT1 affects inflammatory response by targeting miR-129-5p and regulating Notch signaling pathway in epilepsy. Cell Cycle,2020,19(4): 419–431. DOI: 10.1080/15384101.2020.1711578
[19] PENG W X, KOIRALA P, MO Y Y. LncRNA-mediated regulation of cell signaling in cancer. Oncogene,2017,36(41): 5661–5667. DOI: 10.1038/onc.2017.184
[20] SUN J, ZHANG Z, BAO S, et al. Identification of tumor immune infiltration-associated lncRNAs for improving prognosis and immunotherapy response of patients with non-small cell lung cancer. J Immunother Cancer, 2020, 8 (1): e000110[2022-01-14]. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7057423/. doi: 10.1136/jitc-2019-000110.
[21] XUE L, WU P, ZHAO X, et al. Using immune-related lncRNA signature for prognosis and response to immunotherapy in cutaneous melanoma. Int J Gen Med,2021,14: 6463–6475. DOI: 10.2147/IJGM.S335266
[22] TANG R, ZHANG Y, LIANG C, et al. The role of m6A-related genes in the prognosis and immune microenvironment of pancreatic adenocarcinoma. PeerJ, 2020, 8: e9602[2022-01-14]. https://peerj.com/articles/9602/. doi: 10.7717/peerj.9602.
[23] DING H, ZHANG L, ZHANG C, et al. Screening of significant biomarkers related to prognosis of cervical cancer and functional study based on lncRNA-associated ceRNA regulatory network. Comb Chem High Throughput Screen,2021,24(3): 472–482. DOI: 10.2174/1386207323999200729113028
[24] CHEN Q, HU L, HUANG D, et al. Six-lncRNA immune prognostic signature for cervical cancer. Front Genet, 2020, 11: 533628[2022-01-14]. https://doi.org/10.3389/fgene.2020.533628.
[25] XU Y, LUO H, HU Q, et al. Identification of potential driver genes based on multi-genomic data in cervical cancer. Front Genet, 2021, 12: 598304[2022-01-14]. https://doi.org/10.3389/fgene.2021.598304.
[26] LIU Z, WU M, SHI H, et al. DDN-AS1-miR-15a/16-TCF3 feedback loop regulates tumor progression in cervical cancer. J Cell Biochem,2019,120(6): 10228–10238. DOI: 10.1002/jcb.28307
[27] JIANG N, ZHANG X, HE Y, et al. Identification of key protein-coding genes and lncRNAs in spontaneous neutrophil apoptosis. Sci Rep,2019,9(1): 15106. DOI: 10.1038/s41598-019-51597-9
[28] AALIJAHAN H, GHORBIAN S. Long non-coding RNAs and cervical cancer. Exp Mol Pathol,2019,106: 7–16. DOI: 10.1016/j.yexmp.2018.11.010
[29] ZENG J, MA Y X, LIU Z H, et al. LncRNA SNHG7 contributes to cell proliferation, invasion and prognosis of cervical cancer. Eur Rev Med Pharmacol Sci,2019,23(21): 9277–9285. DOI: 10.26355/eurrev_201911_19420
[30] ZHANG G, FAN E, ZHONG Q, et al. Identification and potential mechanisms of a 4-lncRNA signature that predicts prognosis in patients with laryngeal cancer. Hum Genomics,2019,13(1): 36. DOI: 10.1186/s40246-019-0230-6
[31] NAUMANN R W, HOLLEBECQUE A, MEYER T, et al. Safety and efficacy of nivolumab monotherapy in recurrent or metastatic cervical, vaginal, or vulvar carcinoma: Results from the phase I/II CheckMate 358 Trial. J Clin Oncol,2019,37(31): 2825–2834. DOI: 10.1200/JCO.19.00739
[32] KWOK G, YAU T C, CHIU J W, et al. Pembrolizumab (Keytruda). Hum Vaccin Immunother,2016,12(11): 2777–2789. DOI: 10.1080/21645515.2016.1199310
[33] KIM H D, SONG G W, PARK S, et al. Association between expression level of PD1 by tumor-infiltrating CD8+ T cells and features of hepatocellular carcinoma. Gastroenterology,2018,155(6): 1936–1950.e17. DOI: 10.1053/j.gastro.2018.08.030
-
期刊类型引用(2)
1. 尚美芳,李娜,闫佳乐. N6-甲基腺嘌呤相关长链非编码RNA对卵巢癌患者预后的预测价值及免疫功能分析. 癌症进展. 2024(07): 762-770 .  百度学术
百度学术
2. 柳秋霞,嵇龙飞,胡燕勤,潘楠楠,强鑫华,李勇. 血浆外泌体淋巴细胞白血病缺失基因1水平对表皮生长因子受体/间变性淋巴瘤激酶野生型进展期非小细胞肺癌患者免疫治疗反应的预测价值. 中国医药导报. 2023(32): 28-32 .  百度学术
百度学术
其他类型引用(0)

 首页
首页


 下载:
下载: